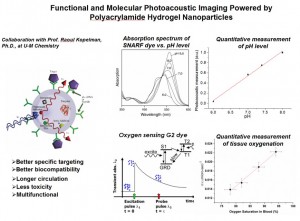
Introduction:
Tumors have different chemical environments (e.g. pH and oxygen) compared to normal tissue. Such chemical concentration differences can be utilized for (1) detecting the tumor, (2) deciding on the appropriate course of therapy, and (3) for monitoring its progress, before, during and after therapy. So far, truly quantitative functional imaging with satisfactory accuracy and resolution has not been achieved in vivo.
Through the collaboration with Prof. Kopelman at UM Chemistry, we are developing a tumor-targeted, biocompatible, biodegradable and bio-eliminable hydrogel-based nanoparticles as contrast agents that enable a combination of structural and functional photoacoustic imaging (PAI). The nanoparticle sonophores, with a size of 30-70 nm, can deliver large payloads to specific cellular targets, enabling PAI with a resolution of 150 μm (spatial), 100 msec (temporal) and a sensitivity of 5 Torr for Oxygen and of 0.1 pH units. More importantly, with flexible engineerability, these nanoparticles have the ability to load multiple agents, not only for multi-analyte diagnosis but also for drug delivery, thus possessing simultaneous diagnostic and therapeutic applicability. This new technology can be used for early detection and diagnosis of cancer, as well as for monitoring the progression of disease and response to therapy. It could also observe phenomena at the molecular level in vivo and allow a better understanding of the pathophysiology of diseases.